The Nuclear Magnetic Resonance (NMR) Facility at the Pontificia Universidad Catolica del Peru initiated its activities in 2003. The first instrument, a Bruker AC300 300 MHz, was upgraded to a Bruker UltraShield 300 MHz with a AVANCE III console in 2010. In 2015 to a Bruker Ascend 500 MHz with AVANCE III HD 500 MHz equipped with an auto-sampler, a Bruker TCI 1h/13C/15N CryoProbe, and a broad.band BBO was acquired. The facility is managed by Prof. Dr. Helena Maruenda (Organic/Analytical Chemistry) and Dr. Juan Lopez (Biomolecular NMR).
The instrument is dedicated to routine organic chemistry analysis and to more specialised studies in the area of Food and Health Sciences, which include metabolomics of natural products and biological fluids, structural dilucidation of macromolecules, implementation of NMR-methologies for drug screening and for evaluation of drug-resistance, among others.
Our fields of research
Analytical chemistry/Metabolomics
Metabolite profiling based on NMR has become a widely applied analytical tool for the identification and discrimination of fruits and other agricultural products according to species, origin, and ripeness, among others. Our interest focusses on different scenarios associated with the Peruvian agroindustry. The discrimination of plants where phenotypic variations are not evident morphologically, the exploration of Vanilla species from the Peruvian amazon as a potential source of new vanilla essence, the determination of optimal roasting conditions of specialty coffees, and the quantitation of carmine in cochineal extracts, are a few examples of studies being conducted in our laboratory in collaboration with several local companies and organizations.
Organic synthesis/ Natural products 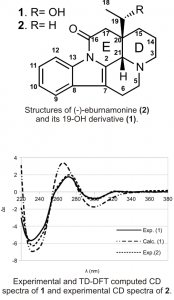
Trypanothione reductase (TryR) is a key enzyme in the metabolism of Trypanosoma cruzi, the parasite responsible for Chagas disease. The available repertoire of TryR inhibitors relies heavily on synthetic substrates of limited structural diversity, and less on plant-derived natural products. Our research is centered in searching for new lead natural product skeletons as inhibitors of TryR from the Peruvian flora. The information obtained from the in vitro and in silico evaluation of the potential leads against TryR, with no in vitro and in silico affinity for human glutathione reductase, is used to optimize the binding mode of the natural product lead through convenient chemical derivatization. Residual dipolar couplings enhanced NMR, in collaboration with Dr. R. Gil, are used to assign the absolute configuration of promising natural product leads.
Bioanalytical chemistry/optical biosensors
Novel methods in food and health sciences based on sensing devices associated with antibodies and oligonucleotides molecular probes are the focus of this line of research. The main objective is to improve the scope of traditional separative methods without compromising sensitivity and selectivity. The optical properties of metallic nanostructures, combined with vibrational spectroscopy or UV-Vis-NIR light are being used to analyze biomolecular systems as well as to quantify the presence toxins in agricultural products. We employ NMR to characterize the binding recognition between molecular probe and analyte.
Members
 Dr. Helena Maruenda (Group Leader)
Dr. Helena Maruenda (Group Leader)
Collaborators
 Dr. Guy Lippens
Dr. Guy Lippens
Publications of members
- Lopez, J. M., Zimic, M., Vallejos, K., Sevilla, D., Quispe-Carbajal, M., Roncal, E., Rodríguez, J., Rodríguez, J., Antiparra, R., Arteaga, H., Gilman, R., Maruenda, H., Sheen, P. (2023) Quantitative 1H-NMR assay from sputum samples for a rapid detection of pyrazinamide resistance in Mycobacterium tuberculosis. JCM (accepted).
- Lopez, J. M., . Leyva, V. E., Bustos, J.A., Perez, E., Sanchez, S., Saavedra, H., Gonzalez, I., Maruenda,H., Garcia. H.H. (2022) Urine 1H-NMR metabolomics to discriminate neurocysticercosis patients from healthy controls. An exploratory study. Am. J. Trop. Med. Hyg. 107,1, pp. 143–145. doi:10.4269/ajtmh.22-0045.
- Lopez, J. M. & Maruenda, H. (2022) Measuring the 3J HNHa coupling by a Simple 2D-intra-HNCA IP/AP-E.COSY with simultaneous encoding of 15N chemical shift and 1J HaCa evolution. Journal of Magnetic Resonance,
- Lopez, J. M., Leyva, V., Maruenda, H. (2021) Pure Shift Nuclear Magnetic Resonance: a New Tool for Plant Metabolomics. J. Vis. Exp. (173), e62719. https://doi:10.3791/62719
- Leyva, V.; Zevallos. A.; Cañar. C; Cabrera, R., Toubiana, D, Lopez, J. M., Maruenda, H. (2021) NMR-based leaf metabolic profiling of V. planifolia and three endemic Vanilla species from the Peruvian Amazon. Food Chem 129365 https://doi.org/10.1016/j.foodchem.2021.129365
- Toubiana, D.; Maruenda, H. (2021) Guidelines for correlation coefficient threshold settings in metabolite correlation networks exemplified on a potato association panel. BMC BioInfromatics Journal.
- Leyva, V, and Maruenda, H. (2021) Assessment of the presence of lipophilic phycotoxins in scallops (Argopecten purpuratus) farmed along Peruvian coastal waters. Journal of Food Protection, 84, 2, 204-212. https://doi.org/10.4315/JFP-20-212.
- Lopez, J.M., Sanchez, L., Nakamatsu, J., Maruenda, H. (2020) Study of the acetylation pattern of Chitosan by pure shift NMR. Analytical Chemistry, 92, 18, 12250–12256. https://doi.org/10.1021/acs.analchem.0c01638.
- Toubiana, T., Cabrera, R., Salas, E., Maccera, Ch., Franco dos Santos, G., Cevallos, D., Lindqvist-Kreuze, H., Boeck, B., Lopez, J.M., Maruenda, H. (2020) Morphological and metabolic profiling of a tropical‐adapted potato association panel subjected to water recovery treatment reveals new insights into plant vigor The Plant Journal, 103: 2193-2210. https://doi.org/10.1111/tpj.14892
- Vallejos-Sánchez, K., Lopez J.M., Antiparra R., Toscano E., Saavedra H., Amzel, M., Gilman, R., Maruenda, H, Sheen, P., Zimic, M. (2020) Mycobacterium tuberculosis ribosomal protein S1 (RpsA) and variants with truncated C-terminal end show absence of interaction with pyrazinoic acid. Scientific Reports, 10 (1), 1-11. https://doi.org/10.1038/s41598-020-65173-z
- Lopez, J.M., Cabrera, R., Maruenda, H. (2019) Ultra-Clean Pure Shift 1 H-NMR applied to metabolomics profiling. Scientific reports 9.1: 6900. https://doi.org/10.1038/s41598-019-43374-5
- Lopez, J.M., Antiparra, R., Lippens, G. Zimic, M; Sheen, P; Maruenda, H. (2018) Backbone chemical shift assignment of macrophage infectivity potentiator virulence factor of Trypanosoma cruzi. Biomol NMR Assign https://doi.org/10.1007/s12104-018-984
- Maruenda H, Cabrera R., Cañari-Chumpitaz, C., Lopez JM., Toubiana D. (2018) NMR-based metabolic study of fruits of Physalis peruviana L. grown in eight different Peruvian ecosystems. Food Chemistry 262: 94-101. http://doi.org/10.1016/j.foodchem.2018.04.032
- Arguelles, A., J.; Cordell, G.A.; Maruenda, H. (2016) Molecular Docking and Binding Mode Analysis of Plant Alkaloids as in vitro and in silico Inhibitors of Trypanothione Reductase from Trypanosoma cruzi. Natural Product Communications, 11, 1, 57 – 62.
- Gant, A.; Leyva, V.E.; Gonzalez, A. E.; Maruenda, H. (2015) Validated HPLC-Diode Array Detector Method for Simultaneous Evaluation of Six Quality Markers in Coffee. Journal of AOAC International, 98, 1, pp. 98-102. http://dx.doi.org/10.5740/jaoacint.14-113.
- Maruenda, H.; Vico, M.L.; Householder, E.J.; Janovec, J. P.; Cañari, C.; Naka, A.; Gonzalez, A.E. (2013) Exploration of Vanilla pompona from the Peruvian Amazon as a potential source of vanilla essence: quantification of phenolics by HPLC-DAD. Food Chemistry 138 161–167. http://doi:10.1016/j.foodchem.2012.10.037
- Trigo-Mouriño, P., Sifuentes, R., Navarro-Vasquez., Gayathri, Ch., Maruenda, H., & Gil, R. R. (2012) Determination of the Absolute Configuration of 19-)H-(-)-eburnamonine Using a Combination od residual Dipolar Couplings, DFT Chemical Shift Predictions, and Chiroptics. Natural Product Communications, No. 6 735-738.
- Householder, E; Janovec, J; Balarezo, A; Huinga, J; Wells, J.; Valega, R. Maruenda, H.; Christeson, E. (2010) Diversity, Natural History, and Conservation of Vanilla (Orchidaceae) in Amazonian Wetlands of Madre de Dios, Peru. J. Bot. Res. Inst. Texas 4 (1) 227-243.
- Galarreta, B., Sifuentes, R., Carrillo, A., Sanchez, L., Amado, M.R.; Maruenda, H. (2008) The Use of Natural Product Scaffolds as Leads in the Search for Trypanothione Reductase Inhibitors. Bioorg. Med. Chem. 16, 6689-6695.
Some additional equipment
- Bruker Esquire 6000 ESI-MS
- Agilent HPLC 1200
- Agilent GC-FID 7890A
- Agilent GC-MS 5977A
- Perkin Elmer LS55 Fluoresence Spectrometer
- Perkin Elmer FRONTIER FTIR (NIR-MIR)
- Perkin Elmer LAMBDA 850 UV-Vis
- FEI Quanta 650 SED-EDX
- Bruker D8-Discover XRD
- Bruker S8-Tiger XRF
- Renishaw´s Raman InVia Reflex
Pontificia Universidad Católica del Perú | Av. Universitaria 1801, San Miguel, Lima 32, Perú
Contact information: hmaruen@pucp.edu.pe









